Research and Development of High Temperature Polymer Electrolyte Membranes and their Application in Fuel Cells
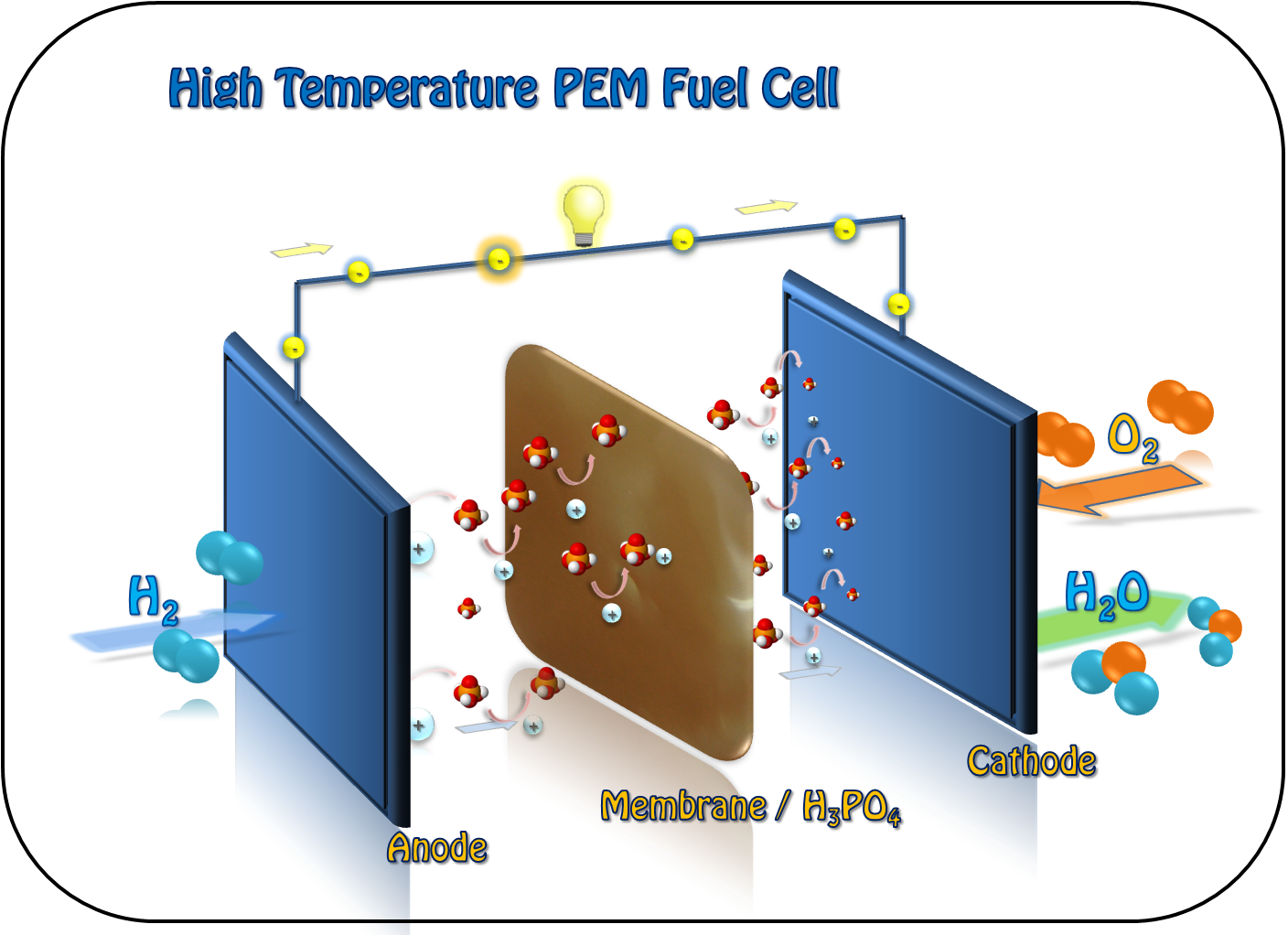
Anode: H2 → 2H++ 2e-
Cathode: 1/2O2 + 2H++ 2e- → H2O
Total Reaction: H2 + 1/2O2 → H2O
Requirments for PEMs
- High Ionic Conductivity
- Electrical Insulator
- Chemical and Oxidative Stability
- Mechanical and Thermal Integrity
- Low gas Permeability
- Low Production Cost
Objective: Development of Novel Polymer Electrolytes for High Temperature PEM-FCs
- Monomer Preparation
- Polymerization via polycondensation
- Characterization via H-NMR, GPC, DMA, TGA, FT-IR, Tensile testing
- Selection of the best membranes for MEA construction and testing
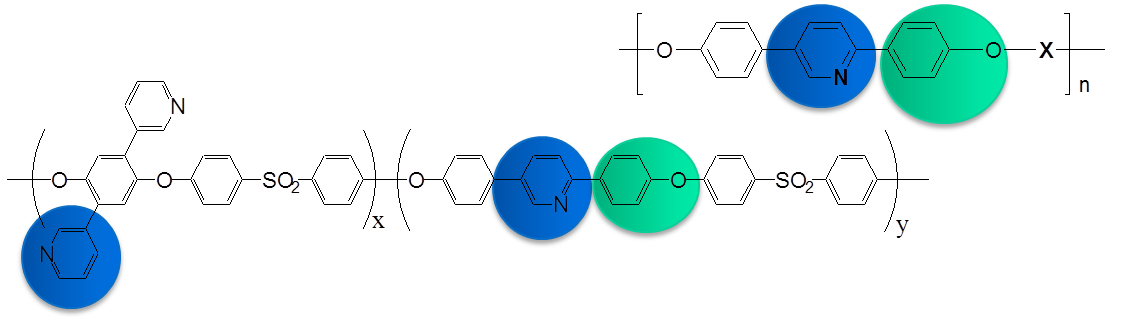
Membranes based on Linear Polymers bearing Polar groups
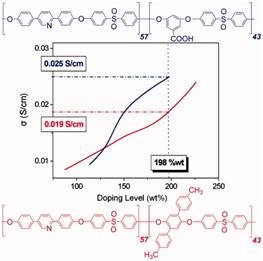
Aromatic polyethers comprised of pyridine main chain units and either phenyl, p-tolyl or carboxyl side ones are presented aiming at high temperature PEMFC applications. The copolymers showed excellent film forming ability, mechanical and thermal properties as well as outstanding oxidative stability even after prolonged exposure under an oxidative environment. The copolymers can be readily doped with phosphoric acid, reaching high doping levels up to 450 wt %. The ionic conductivities obtained are around 0.02 S/cm at RT, while initial single fuel cell tests revealed efficient power output with a current density of 0.5 A/cm2 at 500 mV at elevated temperatures of 160 °C or 180 °C.
Relevant Publications:
- C. Morfopoulou, A. K. Andreopoulou, J. K. Kallitsis, “The Effect of Structural Variations on Aromatic Polyethers for High-Temperature PEM Fuel Cells”, J Polym. Sci.Part A: Polym. Chem. 49, 4325–4334 (2011)
http://onlinelibrary.wiley.com/doi/10.1002/pola.v49.20/issuetoc
Membranes based on Covalently Cross-linked Aromatic Polyethers
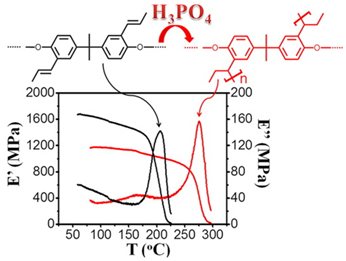
Cross-linkable aromatic polyethers combining main and side chain pyridine units as well as side double bonds were successfully synthesized and characterized. Aiming at the use of these materials as electrolytes at high temperature fuel cells, these polymers were subjected to covalent cross-linking in order to improve their mechanical properties and their stability in the doped state. The cross-linking was obtained via cationic polymerization of the double bonds, during the impregnation of the produced membranes in phosphoric acid. The cross-linking was confirmed by the increased glass transition temperatures, the improved thermal stability and the insolubility of the cross-linked membranes compared to their undoped non cross-linked counterparts. Selected cross-linked membranes were used for membrane electrode assembly (MEA) preparation and tested in a single cell at temperatures between 180 and 220 °C. Long-term durability tests were also performed at 180 °C and at a current density of 0.2 A/cm2. The experiment showed a stable operation without degradation for 1000 h. The promising performance and the durability of the tested materials in combination with the simple and convenient technique which used to produce cross-linked membranes, demonstrates the feasibility of this type of electrolytes to be used in high temperature PEM fuel cell applications.
Relevant Publications:
- K.D. Papadimitriou, M. Geormezi, S. G. Neophytides, J.K. Kallitsis, " Covalent cross-linking in phosphoric acid of pyridine based aromatic polyethers bearing side double bonds for use in high temperature polymer electrolyte membrane fuel cells", J Membr.Sci. 433 1–9, (2013)
http://www.sciencedirect.com/science/journal/03767388/433 - C.I. Morfopoulou, A. K. Andreopoulou, M.K. Daletou, S.G. Neophytides, J.K. Kallitsis, "Cross-linked high temperature polymer electrolytes through oxadiazole bond formation and their applications in HT PEM fuel cells", J Mater. Chem. A: Materials for Energy and Sustainability 1(5), 1613-1622 (2013)
Inside Cover, Volume 49, Issue 20 (pages iii–iv)
Article first published online: 12 SEP 2011 | DOI: 10.1002/pola.25000

The illustration presents the incorporation of a phosphoric acid (H3PO4) doped membrane prepared from one of the aromatic polyether copolymers described by Christina Morfopoulou, Aikaterini K. Andreopoulou, and Joannis K. Kallitsis on page 4325, into a high-temperature polymer electrolyte membrane fuel cell (HT-PEMFC). In this schematic illustration only the two electrodes and the membrane are presented for clarity and simplicity, along with the inlet reactant gases, the transportation of protons along the membrane “hopping” between H3PO4molecules, and the water and electricity produced. Emphasis is given on the polymeric chains bearing polar main chain pyridine and side carboxylic groups.


